How to Find the GFW of a Compound
Here are the three easy steps to finding the GFW of a compound
1) Write down the element and how many times it occurs in the compound
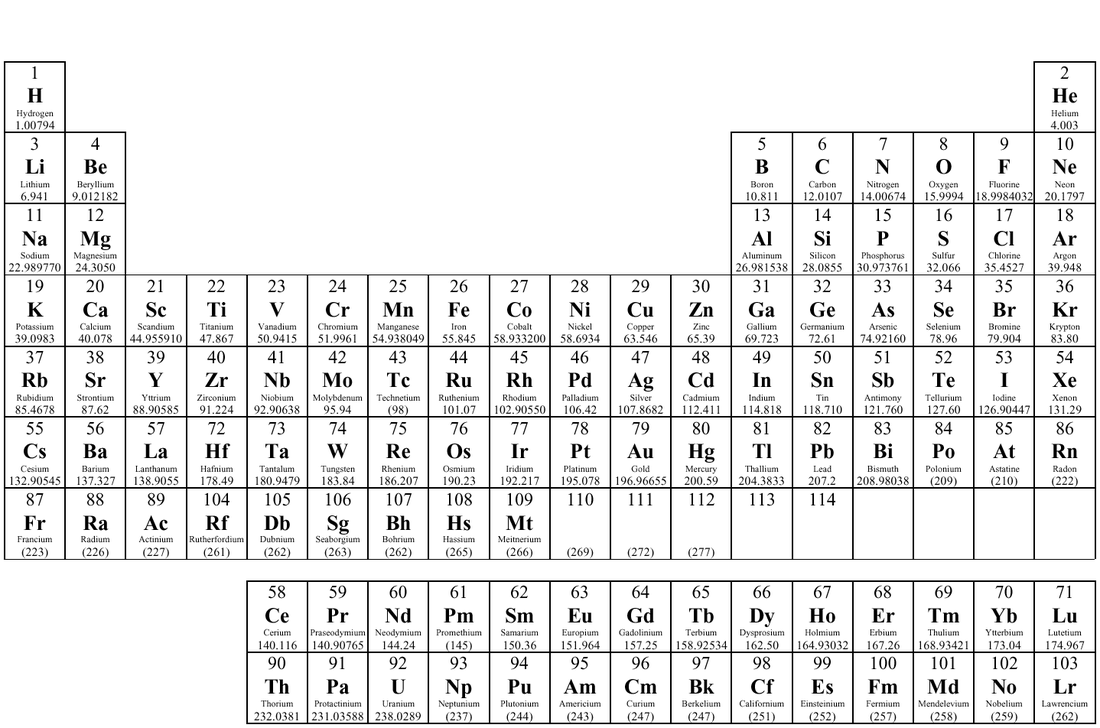
2) Replace the element with its atomic mass (a good website to find the atomic mass is Ptable)
3) Add them all together and you get 98g/mol
1) Write down the element and how many times it occurs in the compound
2) Replace the element with its atomic mass (a good website to find the atomic mass is Ptable)
3) Add them all together and you get 98g/mol
Periodic Table
GFW Example
CaCO3
1. Ca*1, C*1, O*3
2. 40*1, 12*1, 16*3
3. 40+12+48 = 100g/mol
======================
Al(OH)3
1. Al*1, O*3, H*3
2. 27*1, 16*3, 1*3
3. 27+48+3 =78g/mol
======================
Al(NO3)3 =
1. Al*1, N*3, O*6
2. 27*1, 14*3, 16*9
3. 27+42+144 213g/mol
click here to continue learning about how to find the limiting reagent
-or-
click here to go to the limiting reagent quiz
1. Ca*1, C*1, O*3
2. 40*1, 12*1, 16*3
3. 40+12+48 = 100g/mol
======================
Al(OH)3
1. Al*1, O*3, H*3
2. 27*1, 16*3, 1*3
3. 27+48+3 =78g/mol
======================
Al(NO3)3 =
1. Al*1, N*3, O*6
2. 27*1, 14*3, 16*9
3. 27+42+144 213g/mol
click here to continue learning about how to find the limiting reagent
-or-
click here to go to the limiting reagent quiz